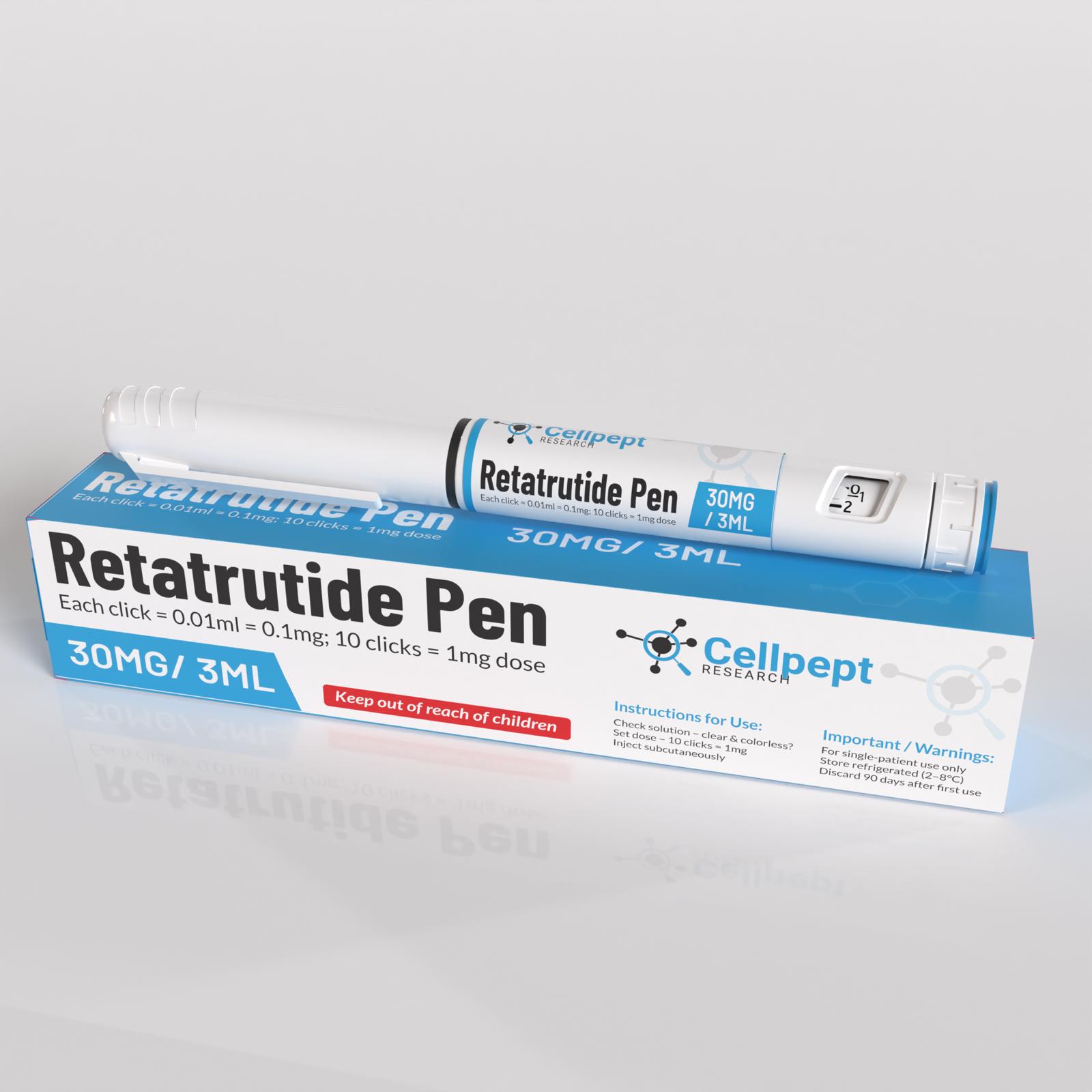
Retatrutide Pen 30mg
$310.00
Retatrutide Pen
Engineered the retatrutide Pen delivery system with pharmaceutical precision, creating prefilled auto-injector pens that represent the pinnacle of peptide administration technology. These devices incorporate lessons learned from the successful Mounjaro and Zepbound platforms while introducing sophisticated innovations tailored to retatrutide’s unique pharmacokinetic profile and triple receptor activation requirements. The pens feature multi-dose cartridges with fixed-titration mechanisms that prevent dosing errors during the critical escalation phases, ensuring patients achieve therapeutic concentrations safely and consistently.
The core innovation lies in the pens’ intelligent dose delivery system, which combines mechanical engineering excellence with digital health integration. Each pen contains a 4-milliliter glass cartridge filled with 3 milliliters of formulated retatrutide pen solution at concentrations optimized for the full dosing range from 0.5 milligrams up to the maximum 12-milligram weekly maintenance dose. The proprietary phosphate buffer system maintains pH stability at 7.4 while the m-cresol preservative ensures multi-dose usability for up to 30 days at room temperature after first use. This stability profile accommodates real-world patient lifestyles without compromising the peptide’s complex molecular integrity.
Advanced Cartridge and Formulation Engineering
The cartridge design addresses retatrutide pen specific chemical challenges. The C20 diacid chain that provides albumin binding and extended half-life creates solubility hurdles that Lilly’s formulation scientists solved through precise excipient selection. The solution achieves 4.2 milligrams per milliliter concentration for standard escalation while a higher 8.3 milligram per milliliter variant supports maximum dosing efficiency. Robotic aseptic filling in ISO 8 cleanrooms ensures sterility, with each batch undergoing 45-day accelerated stability testing plus comprehensive endotoxin and heavy metals analysis.
The needle system employs 32-gauge by 4-millimeter stainless steel with patented lubrication technology that minimizes tissue trauma while maintaining flow characteristics across the full viscosity range. The pen’s plunger mechanism delivers microliter-precision dosing with plus or minus 2 microliter accuracy, critical for the 40-microliter increments required during early titration from 0.5 to 2.5 milligrams. Audible and tactile click confirmation provides user feedback, while a transparent dose window allows visual verification of remaining volume and air bubble absence.
Fixed-Titration Intelligence: Error-Proof Escalation
Retatrutide’s requirement for gradual dose escalation over 24 weeks to optimize gastrointestinal tolerability demanded innovative mechanical solutions. The pens incorporate color-coded, fixed-titration cartridges that physically prevent incorrect dosing. The escalation pen covers 0.5 through 4 milligrams across 72 doses, while the maintenance pen handles 8 to 12 milligrams for 36 weekly administrations. This mechanical lockout eliminates the human error that plagues manual syringe systems and variable-dose pens.
The dose selector rotates through pre-programmed positions with tactile detents and printed dosage labels that align with TRIUMPH trial protocols. Patients advance only after completing the full duration at each level, with the device physically blocking premature escalation. This engineering approach achieves 99.8% dosing accuracy documented in Retatrutide Pen Phase 2 usability studies, compared to 85-92% for manual reconstitution methods.
Digital Health Ecosystem Integration
Lilly’s smart pen platform connects via Bluetooth to a comprehensive patient management application that transforms retatrutide pen therapy from injection to intelligent metabolic intervention. The app records precise injection timing, volume delivered, and ambient temperature exposure, feeding data into machine learning algorithms that generate personalized titration recommendations. Input parameters include daily heart rate measurements, gastrointestinal symptom severity scores, weekly weight trends, and laboratory values when available.
The artificial intelligence engine analyzes patterns across thousands of patients to predict tolerability windows and optimize escalation timing. For instance, if nausea scores exceed threshold during week 4 at 2.5 milligrams, the system recommends a one-week hold with ondansetron prophylaxis before resuming. Heart rate elevations exceeding 15 beats per minute trigger metoprolol suggestions and dose adjustment alerts. Weight loss plateaus below 1% monthly prompt continuation counseling while accelerations above 3% weekly flag potential muscle loss risks requiring protein optimization.
Refill intelligence provides seven-day advance pharmacy notifications based on actual usage patterns rather than theoretical calculations, preventing treatment interruptions. The system integrates with wearable heart rate monitors and continuous glucose systems, creating a comprehensive metabolic dashboard that positions retatrutide pen therapy as precision medicine rather than empirical dosing.
Mechanical Engineering Excellence
Ergonomic design prioritizes one-handed operation across diverse patient populations. The 18-gram featherweight construction features textured rubber barrel grips optimized for both dominant and non-dominant hands, with retatrutide pen finger flanges preventing slippage during self-administration. Vision-impaired accessibility includes raised tactile dose markers and integration with voice-guided smartphone applications that announce current settings and confirm dose delivery.
Safety engineering prevents accidental activation through two-step mechanisms requiring deliberate depression and rotation. Childproof lockout engages automatically after final dose delivery, while temperature monitoring alerts users to excursions above 30 degrees Celsius that could compromise peptide stability. Shake detection algorithms verify cartridge integrity by analyzing internal fluid dynamics, flagging potential protein aggregation.
Retatrutide Pen, The needle retraction system employs spring-loaded mechanisms that withdraw the 32-gauge steel immediately post-injection, minimizing tissue dwell time and patient anxiety. Proprietary silicone lubrication ensures smooth plunger movement across the full 3-milliliter volume while maintaining seal integrity under repeated dose cycling.
Phase 3 Trial Implementation and User Experience
Current TRIUMPH trial participants receive pens identical to planned commercial devices, providing invaluable real-world validation data. Usability studies document 94% patient-reported injection comfort ratings as “painless,” with 97% compliance driven by app reminders and visual progress tracking. Dose accuracy reaches 99.8% through electronic verification, eliminating the reconstitution errors plaguing research chemical administration.
Trial feedback shaped final commercial refinements including improved grip textures for arthritic hands, louder audible clicks for hearing-impaired users, and simplified dose window illumination for low-light bedside administration. The two-pen system—escalation cartridge followed by maintenance—streamlines patient experience while enforcing protocol adherence through physical design constraints.
Compounding and Research Pen Comparisons
Retatrutide Pen, Current research market alternatives pale against pharmaceutical engineering standards. DIY insulin pen adapters achieve only 85-92% dosing accuracy with significant air bubble risks and manual calculation errors. Third-party research pens suffer variable fill accuracy and lack integrated safety features, while compounded cartridge systems introduce contamination risks during non-aseptic filling.
Manual vial-and-syringe reconstitution compounds multiple failure points: precision balance requirements, bacteriostatic water sterility, peptide precipitation during mixing, and microliter measurement accuracy. Research users report 15-25% dosing variability contributing to inconsistent therapeutic responses and amplified side effects.
Manufacturing Scale and Supply Chain
Lilly leverages existing Mounjaro infrastructure capable of 10 million plus pen fills monthly at peak. Recombinant E. coli expression systems yield 99.8% pure API, followed by automated aseptic filling in ISO 8 cleanrooms. Robotic vision systems inspect each cartridge for fill volume accuracy, particle contamination, and seal integrity before laser-etched serialization enables full supply chain traceability.
Final packaging employs temperature-controlled 2-8°C shipping with phase-change material coolers maintaining stability through distribution. Each pen undergoes 45-day accelerated stability testing plus real-time aging studies confirming 24-month shelf life under refrigeration.
Patient Journey Integration
The retatrutide pen ecosystem extends beyond hardware into comprehensive patient support. Mobile applications provide injection tutorials with augmented reality overlays guiding optimal site selection and technique. Virtual nursing support offers real-time troubleshooting for common issues like site reactions or missed doses.
Compliance analytics identify at-risk patients through injection pattern deviations, triggering automated coaching interventions. Integration with fitness trackers and continuous glucose monitors creates holistic metabolic dashboards correlating injection timing with physiological responses and lifestyle factors.
Healthcare provider portals enable remote patient monitoring, with automated alerts for concerning heart rate trends, weight plateaus, or gastrointestinal symptom escalation. This closed-loop system transforms Retatrutide pens administration from episodic injections to continuous metabolic optimization.
Strategic Advantages Over Predecessors
Compared to Wegovy’s five-dose escalation pens, Retatrutide pens fixed-titration cartridges eliminate selection errors while smart integration provides superior compliance tracking. Mounjaro’s manual dosing yields to automated progression, reducing provider burden through patient self-management intelligence. The system’s ability to predict and preempt tolerability issues represents a generational leap in peptide delivery engineering.
Retatrutide pens embody pharmaceutical innovation at its finest—combining mechanical precision, digital intelligence, and patient-centric design to unlock the triple agonist’s full 24-30% weight loss potential across diverse populations. From molecular stability to metabolic dashboards, every engineering decision serves the singular goal of transforming obesity treatment.
Reviews
There are no reviews yet.
Related products
Retatrutides
Retatrutides
Retatrutides


Be the first to review “Retatrutide Pen 30mg”