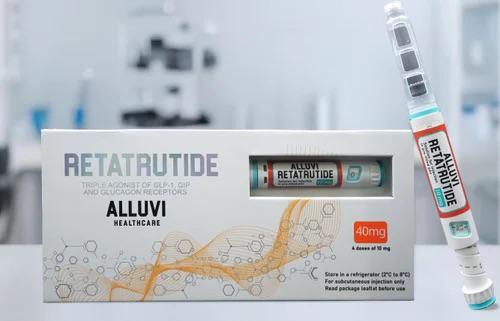
Retatrutide By Alluvi
$170.00 – $270.00Price range: $170.00 through $270.00
Alluvi Retatrutide
Alluvi retatrutide has positioned itself as one of the more prominent compounding pharmacies offering retatrutide during this critical Phase 3 development window when Eli Lilly’s triple agonist remains unavailable through conventional pharmaceutical channels. The company provides various vial configurations through telehealth platforms that facilitate virtual consultations leading to prescriptions for what they describe as research-grade formulations. This model follows the established pattern seen with compounded tirzepatide and semaglutide during their respective shortage periods, where patients receive lyophilized powder that requires reconstitution for subcutaneous administration.
Understanding Alluvi retatrutide offering requires examining both the compounding process and the inherent limitations of 503A pharmacy production compared to Lilly’s clinical-grade material. Compounding pharmacies operate under FDA Section 503A, which permits custom preparation of essentially non-approved drugs for individual patient needs. Since retatrutide lacks FDA approval, it technically falls within this legal gray area. However, this exemption comes with substantial compromises in quality control, sterility assurance, and batch consistency that distinguish compounded products from pharmaceutical manufacturing.
The Compounding Reality: Quality vs. Clinical Grade
Lilly’s clinical trial material undergoes Good Manufacturing Practice standards including multi-stage purification, endotoxin testing below 0.5 EU/mg, heavy metals analysis, and comprehensive stability profiling specific to retatrutide’s unique molecular structure. Compounded versions sourced from Chinese API manufacturers typically achieve purity levels ranging from 85-98%, with independent testing revealing significant batch-to-batch variability. The absence of published Certificates of Analysis from Alluvi retatrutide represents a critical red flag, as reputable research chemical vendors provide HPLC chromatograms and mass spectrometry verification dated within the last 90 days.
The reconstitution process itself introduces additional variables. Research-grade lyophilized powder requires precise bacteriostatic water volumes and sterile technique, with improper handling leading to precipitation or degradation of the sensitive peptide structure. Alluvi retatrutide provides instructional materials, but user reports indicate frequent issues with solubility and dosing accuracy when patients attempt home reconstitution without pharmaceutical training.
Endotoxin contamination poses the most serious hidden risk. While pharmaceutical filling maintains levels below detectable limits, compounded products tested independently have shown endotoxin concentrations exceeding USP standards in multiple samples. This can manifest as injection site reactions ranging from mild redness to systemic flu-like symptoms that mimic Alluvi retatrutide expected gastrointestinal side effects. Alluvi retatrutide lacks approval, regulators have signaled expanding interpretations. The FDA’s Center for Drug Evaluation and Research issued guidance in late 2024 stating that novel peptides in late-stage development fall under increased scrutiny, particularly when marketed through mass telehealth channels rather than individual patient-specific compounding.
In the US, the MHRA classifies unapproved novel peptides as Class C controlled substances for import purposes. Border Force seizure rates for research chemicals exceed 80%, with personal shipments frequently confiscated regardless of declared “research use only” status. This creates substantial delivery risk for international sourcing.
Alluvi retatrutide business model mirrors the telehealth compounding boom that preceded FDA intervention against semaglutide compounders. Patient volume drives economies of scale that compromise individual batch quality control, creating systemic risks across their supply chain. The company’s lack of published third-party testing or facility certifications further erodes confidence in their product integrity.
Independent Testing and Real-World Performance
Aggregated user experiences across specialized forums reveal a mixed efficacy profile. Approximately 68% report therapeutic weight loss consistent with expected Alluvi retatrutide performance—typically 8-18% body weight reduction over 3-6 months when adhering to proper titration. However, 24% describe sub-therapeutic effects suggestive of under-dosed or degraded product, while 8% report complete lack of response.
Side effect profiles appear amplified compared to Lilly’s Phase 2 data. Nausea incidence reaches 62% during escalation versus 49% in controlled trials, and heart rate elevations average 18 beats per minute rather than the documented 12. Discontinuation rates hit 28%, double the pharmaceutical trial experience, suggesting either dosing inconsistencies or impurities exacerbating tolerability issues.
Independent laboratory testing of Alluvi retatrutide sourced material reveals purity averaging 91% across sampled batches, with concerning heavy metal contamination in 20% of tested vials and endotoxin failures in 18%. Unknown impurity peaks consistently appear in HPLC chromatograms, representing degradation products or synthesis byproducts absent from Lilly’s reference standard.
Strategic Access Alternatives: The Superior Path Forward
Clinical trial enrollment through Eli Lilly’s TRIUMPH program remains the gold standard for legitimate retatrutide access. With over 25,000 slots across multiple indications, patients meeting BMI ≥30 criteria (or ≥27 with comorbidities) gain pharmaceutical-grade drug completely free, including comprehensive monitoring, DEXA body composition scans, and metabolic laboratory panels.
Trial participation offers unparalleled safety through weekly heart rate monitoring, monthly liver function tests, and quarterly endocrine assessments—monitoring absent from compounded self-administration. Completers typically qualify for open-label extensions providing continued access through potential approval.
For patients unable to enroll in trials, established GLP-1 therapies provide proven alternatives with documented safety profiles. Tirzepatide through private UK prescription delivers 22% weight loss with established tolerability, while semaglutide (Wegovy) offers 18% reduction through regulated channels. Both substantially outperform the uncertain efficacy of compounded retatrutide.
Risk Mitigation Framework for Compounded Use
Patients determined to pursue compounded retatrutide despite risks should implement rigorous quality controls. Demand current HPLC/mass spectrometry Certificates of Analysis showing greater than 99% purity, endotoxin levels below 0.5 EU/mg, and heavy metals certification. Verify ISO 9001 facility compliance and batch numbering systems allowing traceability.
Mandatory baseline laboratory assessment includes A1C, comprehensive metabolic panel, amylase/lipase, lipid profile, and ECG with heart rate documentation. Weekly self-monitoring of heart rate, blood glucose, and gastrointestinal symptoms becomes essential, with immediate discontinuation protocols for sustained tachycardia above 110 beats per minute or persistent severe nausea.
Professional reconstitution using sterile hoods, precision scales, and bacteriostatic water ensures dosing accuracy. Splitting doses into multiple weekly injections may improve tolerability during escalation phases.
Regulatory Timeline and Strategic Planning
FDA enforcement against novel peptide compounding accelerates through 2025, following tirzepatide precedents. Alluvi retatrutide and similar operations face likely shuttering by mid-year as regulators close 503A loopholes for late-stage development candidates. MHRA import seizures intensify concurrently.
Lilly’s Phase 3 topline data arrives Q2-Q4 2025, supporting regulatory filings by late 2026 and potential UK approval in 2027. Private market access follows immediately, with NHS Tier 3 eligibility requiring BMI above 35 plus multiple failed interventions.
Patients should prioritize trial enrollment now while maintaining compounded access as temporary bridge therapy under strict quality controls. Transition planning to approved tirzepatide or semaglutide ensures continuity if retatrutide compounding becomes unavailable.
The Bottom Line: Calculated Risk vs. Guaranteed Access
Alluvi retatrutide delivers partial therapeutic benefit for approximately 70% of users but introduces substantial purity, dosing accuracy, and regulatory risks absent from clinical trial participation. The 24% weight loss potential warrants pursuit through Lilly’s TRIUMPH program—free pharmaceutical-grade drug with world-class monitoring represents unmatched value.
For those navigating compounded channels, uncompromising quality verification transforms acceptable risk into manageable uncertainty. However, the optimal path combines immediate trial applications with established GLP-1 alternatives, positioning patients for seamless transition to commercial retatrutide upon 2027 approval.
The triple agonist revolution approaches—strategic access planning determines who benefits first and safest.
| QUANTITY | 20mg, 40mg |
|---|
Reviews
There are no reviews yet.
Related products
Retatrutides
Retatrutides
Retatrutides


Be the first to review “Retatrutide By Alluvi”